Table of Contents
Get started with MyPerfectResume today!
- Build a resume on any device
- Pick an ATS-friendly template
- Tailor with AI copy suggestions
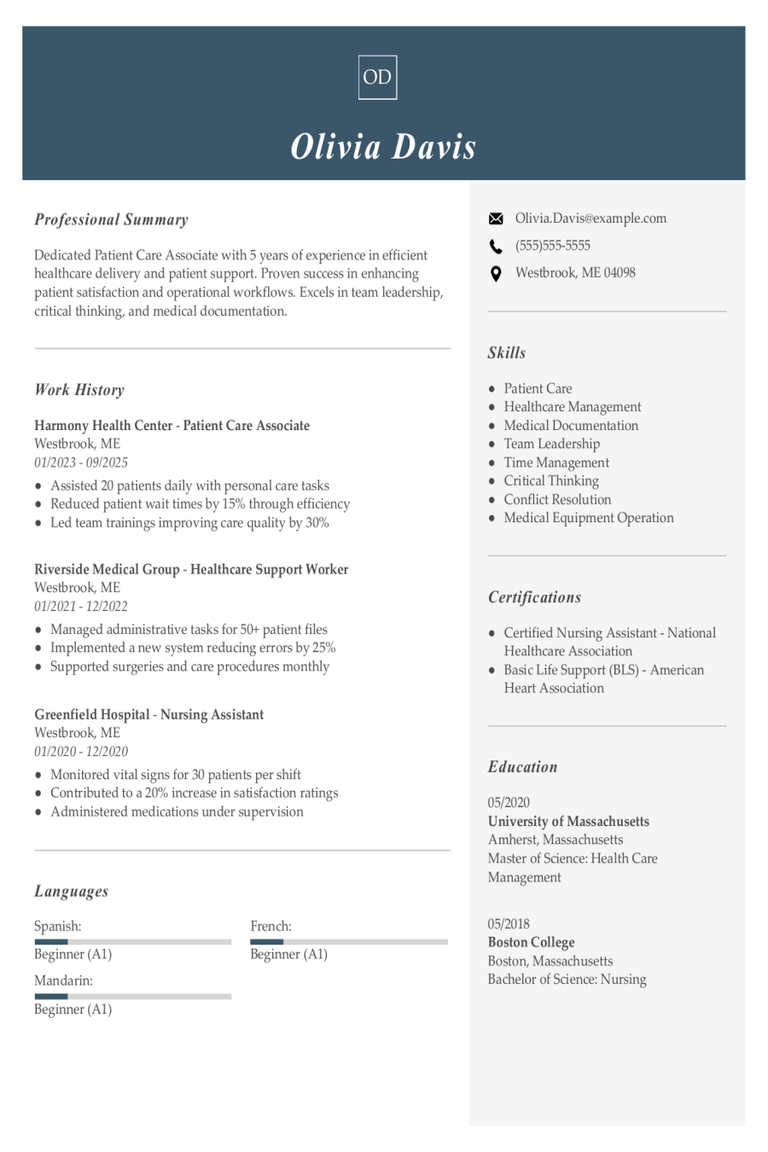
Why this resume works
- Quantifies accomplishments: Measurable accomplishments, like a 30% boost in participant recruitment, highlight the applicant’s impressive contributions to clinical trial success.
- Uses action-oriented language: Through action verbs such as “managed” and “implemented,” the job seeker reflects proactive involvement and strong leadership in research settings.
- Highlights industry-specific skills: Featuring skills like protocol development and regulatory compliance, the applicant showcases their healthcare skills essential for thriving in clinical research environments.
More Clinical Trial Associate Resume Examples
Explore our clinical trial associate resume examples to effectively showcase your project coordination, regulatory compliance, and data management skills. Use these medical resume samples to create a resume that highlights relevant experience in clinical research settings.
Entry-Level Clinical Trial Associate
Why this resume works
- Centers on academic background: The applicant’s master’s degree in clinical research management lays a strong educational foundation for their career.
- Effective use of keywords: By including role-specific terms like “compliance auditing” and “clinical trial management,” the applicant uses keywords to improve ATS compatibility.
- Puts skills at the forefront: This resume prioritizes important skills such as data analysis and protocol development, aligning with the skills-based resume format strategy that benefits new professionals.
Mid-Level Clinical Trial Associate
Why this resume works
- Includes a mix of soft and hard skills: Combining technical skills like clinical trial management with interpersonal skills, the applicant effectively coordinates teams and improves patient recruitment.
- Points to measurable outcomes: By reducing trial setup time by 20% and increasing patient recruitment by 30%, the applicant showcases a knack for delivering effective, measurable results in research settings.
- Demonstrates language abilities: The applicant’s ability to communicate in Spanish, French, and German highlights language skills important for international collaborations in clinical trials.
Experienced Clinical Trial Associate
Why this resume works
- Focuses on work history: Opting for a chronological resume format, the applicant effectively showcases their extensive career growth, detailing each role’s impact on clinical research advancements over time.
- Lists relevant certifications: By listing comprehensive certifications like CCRC and Clinical Data Management Certification, the applicant highlights ongoing dedication to improving their clinical research expertise.
- Emphasizes leadership skills: Demonstrating strong leadership skills, the applicant managed participant recruitment and coordinated trials across multiple sites, driving team success in complex environments.
Clinical Trial Associate Resume Template (Text Version)
Use the template below, or browse our resume templates for a wider variety of designs.
Chris Huang
Indianapolis, IN 46203
(555)555-5555
Chris.Huang@example.com
Professional Summary
Clinical Trial Associate skilled in optimizing trial operations, enhancing participant engagement, and ensuring data integrity through effective project management strategies.
Work History
Clinical Trial Associate
Innovate Biopharma – Indianapolis, IN
June 2023 – August 2025
- Managed 5 clinical study sites
- Increased participant recruitment by 30%
- Streamlined data entry processes
Research Coordinator
Natura Labs – Indianapolis, IN
June 2019 – May 2023
- Implemented new quality controls
- Decreased data errors by 15%
- Collaborated on study protocols
Clinical Research Assistant
MediSync Trials – Indianapolis, IN
June 2017 – May 2019
- Supported over 12 clinical studies
- Contributed to participant screening
- Maintained accurate trial databases
Languages
- Spanish – Beginner (A1)
- French – Beginner (A1)
- German – Beginner (A1)
Skills
- Clinical Trial Management
- Data Analysis
- Patient Recruitment
- Regulatory Compliance
- Protocol Development
- Project Coordination
- Quality Assurance
- Medical Terminology
Certifications
- Certified Clinical Research Associate – Association of Clinical Research Professionals
- Clinical Trial Management Certification – Society of Clinical Research Associates
Education
Master of Science Clinical Research
Harvard University Cambridge, Massachusetts
May 2017
Bachelor of Arts Biology
University of California, Los Angeles Los Angeles, California
May 2015
Related Resume Guides
- Clinical Documentation Improvement Specialist
- Clinical Project Manager
- Clinical Research Assistant
- Coding Specialist
- Dermatologist
- Dialysis Technician
- Doctor
- Electrocardiograph Technician
- Emergency Department Technician
- Emergency Room Registration Clerk
- Equipment Technician
- Health Information Technician
- Hospital Administrator
- Hospital Volunteer
- Intake Coordinator
- Intensive Care Physician
- Medical Laboratory Technologist
- Medical Records Specialist
- Medical Representative
- Medical Research Assistant
- Midwife
- MRI Technologist
- Neurosurgeon
- Nuclear Medicine Technologist
- OB-GYN Medical Assistant
- Ophthalmologist
- Ophthalmology Assistant
- Optometrist
- Orthopedic Surgeon
- Pediatric Assistant
- Pediatrician
- Physician
- Physician Assistant
- Podiatrist
- Psychiatrist
- Radiologist
- Radiology Technician
- Resident Medical Officer
- Respiratory Therapist
- Speech Language Pathologist
- Surgeon
- Surgery Scheduler
- Ultrasound Technician
Advice for Writing Your Clinical Trial Associate Resume
Explore our advice section on how to write a resume tailored for a clinical trial associate role. Discover how to highlight your skills in managing trials, collaborating with research teams, and ensuring compliance with protocols. Whether you’re new to the field or looking to move up, these tips can help you create a standout resume that captures your expertise and passion for clinical research.

Highlight relevant certifications and training
Certifications and specialized training are important for a clinical trial associate because they show you have the knowledge and skills needed to help in medical research. Adding these credentials to your resume can include learning about regulations, understanding trial design, or handling data.
They ensure you know how to do your job well and follow rules that keep trials safe and reliable. Having these certifications on your resume shows employers you’re ready to support their research effectively. Here are a few examples of certifications for your resume:
- Clinical Research Coordinator (CRC) Certification
- Good Clinical Practice (GCP) Training
- Certified Clinical Research Professional (CCRP)
- Human Subjects Protection Training
- Data Management Certification
These certifications prove you can handle specific tasks required in clinical trials. This preparation helps ensure successful project outcomes.
Example of a certifications & training section
Good Clinical Practice (GCP)
Issued by: National Institute of Health (NIH)
Issued 2023
Clinical Research Coordinator (CRC)
Issued by: Association of Clinical Research Professionals (ACRP)
Expires 2024
Biomedical Data Analysis
Issued by: Stanford Online
Completed 2022
Pharmaceutical Drug Development
Issued by: University of California, San Diego Extension
Completed 2021
Ethics in Clinical Trials
Issued by: CITI Program
Expires 2025
Looking at professional resume examples can give you ideas on how to make your own resume look great and stand out.
List your most relevant skills
For a clinical trial associate, it’s important to balance both technical and soft skills. Technical skills include knowing software like Microsoft Office, Electronic Data Capture (EDC) systems, and Clinical Trial Management Systems (CTMS). Understanding data entry, regulatory documents, and trial protocols is also key. These skills help ensure that the trials run smoothly and all data is accurately recorded.
Soft skills are just as important. Being detail-oriented helps catch errors before they become problems. Good communication skills make it easier to work with different teams and explain complex ideas simply. Time management ensures that tasks are completed on schedule, which is important in clinical trials.
Include a skills section on your resume to highlight these abilities right away. Mention your software knowledge and any relevant artistic talents here, too. Also, weave these skills into your work experience bullet points for more impact.
For example, instead of just saying you managed data entry, mention how your attention to detail helped maintain accurate records or how your time management kept the project on track. This shows you can apply your skills effectively in real-world situations.
Choose a resume format that highlights your experience with clinical trials, adherence to timelines, and collaboration skills to make a strong impact as a clinical trial associate.
Write a strong professional summary
A professional summary on a resume serves as an introduction to hiring managers, helping them quickly understand who you are and what you bring to the table. It gives readers the choice between highlighting past achievements with a summary or focusing on future goals with an objective.
A professional summary is three to four sentences that showcase your experience, skills, and accomplishments. It’s best for those with experience, like a clinical trial associate, as it highlights your professional identity and value to potential employers. This helps paint a clear picture of what you’ve accomplished in your career.
In contrast, resume objectives are statements about career goals. These are better suited for entry-level applicants, career changers, or those with employment gaps. While a summary focuses on “what I’ve accomplished,” an objective centers on “what I aim to contribute.” Next, we’ll provide examples of both summaries and objectives tailored to different industries and levels of experience.
Clinical trial associate resume summary examples
Entry-level
Newly graduated clinical trial associate with a Bachelor of Science in biology and certification in Good Clinical Practice (GCP). Experienced in data collection during academic projects, familiar with regulatory guidelines, and eager to contribute to clinical research teams. Strong organizational skills and keen attention to detail for managing trial documentation.
Mid-career
Clinical trial associate with over five years of experience supporting phase II and III trials across oncology and cardiology domains. Proven track record in coordinating participant recruitment, monitoring compliance, and maintaining accurate study records. Known for effective communication with cross-functional teams and dedication to advancing robust clinical outcomes.
Experienced
Senior clinical trial associate specializing in complex global trials with expertise in protocol development and site management. Experienced in leading project timelines, ensuring adherence to international regulations, and optimizing operational processes. Committed to driving successful trial execution while mentoring junior associates on best practices within the field.
Clinical trial associate resume objective examples
Recent graduate
Detail-oriented recent graduate with a Bachelor of Science in biology seeking an entry-level clinical trial associate role. Eager to apply strong analytical skills and foundational knowledge in clinical research processes to support the successful execution of clinical trials.
Career changer
Passionate professional transitioning from laboratory science to a clinical trial associate position, bringing robust skills in data analysis and project management. Looking forward to contributing to a collaborative team environment and advancing medical research efforts.
Entry-level
Aspiring clinical trial associate with experience in academic research settings, aiming to contribute careful attention to detail and organizational abilities to a leading clinical research organization. Committed to ensuring compliance and quality standards in all phases of clinical trials.
Use our Resume Builder to make your clinical trial associate resume stand out. It helps you organize your skills and experience quickly with easy-to-use templates.
Showcase your accomplishments and publications
Listing publications, conference presentations, and research accomplishments can make your resume stand out when applying for a clinical trial associate position. These elements show that you have contributed to the scientific community and possess strong research skills.
Including them in a structured publications section helps employers see your expertise and dedication to advancing knowledge in your field. This approach is particularly useful if you’ve worked on studies or trials relevant to the job you’re seeking.
When creating this section, list peer-reviewed articles, book chapters, and conference papers using professional formats like APA or MLA. Include citation details such as journal name, volume, issue, and year to provide clear reference points.
Highlight any major awards or grants you’ve received, as they support your credibility and show recognition from peers. If you have an online research profile on platforms like Google Scholar or ORCID, consider adding links so employers can explore more of your work.
These steps not only improve your resume but also demonstrate your active engagement in ongoing research efforts. They tell potential employers that you are up-to-date with industry standards and committed to making meaningful contributions as a clinical trial associate.
Example of publications section
Publications
- Doe L., Smith J. “Electronic Trial Master File Systems: Enhancing Accuracy and Compliance.” Regulatory Affairs Journal, 2021.
- Doe L., Martinez K. “Best Practices for Clinical Trial Documentation and Compliance.” Journal of Clinical Research Operations, 2023.
- Chen R, Doe L., Patel S. “Optimizing Site Monitoring Visits to Improve Trial Efficiency.” Clinical Operations Review, 2022.
Select a resume template that is simple and neat, with clear headings and sections. Avoid using too many colors or fancy fonts that can be distracting.
FAQ
Do I need to include a cover letter with my clinical trial associate resume?
Including a cover letter with your clinical trial associate resume can boost your application and increase interview opportunities.
It allows you to highlight why you’re specifically interested in the clinical trial associate position and showcase relevant skills like data management or regulatory knowledge.
If the company is recognized for certain trials or research areas, mention any related experience you have to show your enthusiasm and expertise.
Use tools such as our Cover Letter Generator to craft a tailored cover letter that complements your resume, guiding you through each step with expert advice.
Additionally, explore our collection of cover letter examples to find samples that align with your career goals and industry standards.
How long should a clinical trial associate’s resume be?
For a clinical trial associate, a one-page resume is typically sufficient to highlight key skills such as attention to detail, familiarity with regulatory requirements, and experience in managing trial documentation. It’s important to demonstrate your ability to collaborate within a team and your knowledge of clinical protocols.
However, if you have extensive experience or relevant certifications, a two-page resume might be better suited. This gives you space to outline significant achievements and complex projects without cluttering the page.
Ultimately, how long a resume should be depends on your career stage and the depth of your expertise. Focus on including only details that add value to the role you’re pursuing.
How do you write a clinical trial associate resume with no experience?
When crafting a resume with no experience for a clinical trial associate position, focus on highlighting your educational background, relevant skills, and any training or certifications that align with the role. Here are a few tips to help you get started:
- Emphasize your education: Start by listing your degree in life sciences or a related field. Include the name of the institution, graduation date, and any honors or relevant coursework like biostatistics or research methodology.
- Showcase transferable skills: Highlight skills such as attention to detail, organizational abilities, and skill in data management software. If you have experience in research projects during your studies, mention these too.
- Include relevant certifications: If you have completed any courses, like Good Clinical Practice (GCP) certification or other research-related training programs, be sure to list them prominently.
For more tailored guidance on creating a resume with no experience specific to this field, consider consulting resources from career experts or connecting with professionals already working as clinical trial associates.
Rate this article
Clinical Trial Associate
Share this page